-
健康危险行为是对人们的身体健康和精神状态产生危害或潜在危害的行为[1],与情感和认知有密切关系。尤其是青春期的个体,由于身心发展不平衡,更容易卷入危害健康的行为之中,但目前对青少年健康危险行为的研究主要是行为学的研究。文献[2]对6 633名青少年的研究中发现,在所有的危险行为上有较高概率的高危险组占全体人员的13.6%,这对青少年的身心健康乃至成年后的生活造成了严重的伤害。实证研究发现,不同形式的健康危险行为(比如暴力攻击、吸毒、违反纪律等)常常伴随发生[3],为了解释这种现象,文献[4]提出了危险行为理论,认为各种不同形式的危险行为是由潜在的共同问题行为因子决定的。这种潜在的问题行为因子是否跟大脑的网络组织有关呢?可能跟哪些脑区有关,脑区间有怎样的相互作用,以及又有怎样的脑网络特征?本文拟通过利用动态功能连接对健康危险性行为特征的预测来研究与青少年健康危险行为相关的脑网络模式。
在目前有关功能连接的研究中,功能连接主要是通过计算在整个静息态fMRI扫描时程中各脑区的时间序列相关性得到,称之为静态功能连接[5]。然而最近研究表明在静息态下的功能连接也是动态变化的,显示出显著的振幅波动性[6],而且在静息态下观测到的低频振荡呈现出复杂的空时结构,包含多个离散的稳定模式[7]。因此,研究认为静息态下功能连接的动态性反应了与认知和行为能力相关的脑区间动态的相互作用[8]。猜想这种动态的相互作用可能与青少年的健康危险行为相关。因此,本文的研究通过使用基于支持向量机(SVM)的回归分析(SVR)利用动态功能连接对个体的健康危险行为评分进行预测,以实现对青少年健康危险行为的脑网络研究。
-
为获得健康危险行为比较严重的对象,本文从湖南省少年犯管教所招募已到法定年龄的男性志愿者23名(进入时未到法定年龄),并同时在社区召募了30名年龄和教育程度匹配的健康对象一起进行研究。所有的参与人员均为右利手,至少半年没有吸毒或接触酒精,也没有精神类疾病(比如抑郁症,焦虑症和精神分裂症),年龄在18~23岁,教育年限6~9年,智商正常。
实验前向所有参与人员告知实验的相关过程及可能的不适,然后签署知情同意书。本研究经过湖南省少年犯管教所和中南大学湘雅三医院伦理委员会同意。
-
为了测量每一个对象的健康危险性行为特征分数,本文使用了HBICA量表(the health-risk behavior inventory for Chinese adolescents)[2]。HBICA量表是一个自我报告式的问卷调查,一共包含33个相关条目,每一个条目的内容用1~5分进行打分,1分表示该种行为从来没有,5分表示该种行为经常出现。总分在33~155的范围内,分数越高表明危险性行为的水平越高。HBICA量表能有效地检测青少年的危险性行为特征并有效的辨别出高危险性的个体,有着良好的信度(0.66~0.76)和效度(0.77~0.86) [2]。
-
所有参与者的静息态fMRI数据均使用中南大学湘雅三医院的Philips磁共振成像3T系统得到。扫描前,告知被试闭眼、放松,保持清醒,不要进行特定的认知活动,头部保持不动。扫描后询问被试是否在扫描中睡着,当时在想什么,来证实被试是否满足上述条件。使用梯度平面回波成像(EPI)序列进行扫描,相关参数如下:TR:2 000 ms,TE:30 ms,FOV:240 mm,层数:36,层厚:4.0 mm,无间隙,翻转角:90°,扫描矩阵128×128。扫描时程为400 s共得到200幅全脑图像。
使用SPM8对静息态fMRI数据进行预处理:考虑到磁场饱和的时间,去掉每个被试的前5幅图像,对剩下的195幅图像进行空间校正和头动校正,去掉三维平移超过1 mm、三维旋转超过1°的4个被试;然后空间标准化到EPI模板(MNI坐标),重采样为3×3×3的体素大小;再进行空间平滑和时间滤波,分别使用8 mm半高全宽的高斯核函数和Chebyshev带通滤波器(0.01 < f < 0.1Hz);最后对几个无关的变量进行回归,包括头动参数、全脑平均信号、白质信号和脑脊液信号,以去除一些生理噪声在功能连接分析中的潜在影响[9]。
-
按照功能把大脑分成160个感兴趣脑区(ROI)[10],根据此模板,本文求取动态功能连接网络。首先对每个对象预处理后的全脑图像计算每个ROI脑区内全部体素的平均时间序列,然后计算在滑动窗为40 s时任意两个ROI脑区信号的Pearson相关系数,得到176个对称的相关系数矩阵(160×160)表示的动态功能网络,去除矩阵对角线上的元素,提取矩阵的下三角元素并进行Fisher’s Z-变换使其标准化,得到一个12 720维相关系数向量。最后求取相关系数时间序列的波动振幅(ALFF-FC),ALFF-FC表示了每一条连接的动态特性,之前的研究表明ALFF-FC能很好的度量信号的波动,被定义为在低频范围内整体的信号能量[7, 11]。为了去除由于滑动窗的使用而引入的冗余波动,本文根据滑动窗大小w对得到的相关系数时间序列进行1/w的低通滤波,再应用快速傅里叶变换(FFT),并对FFT系数求和从而得到从0到1/w的动态功能连接频段内的ALFF-FC指数[12]。相关系数向量的ALFF-FC值反应了全脑动态功能连接网络的变化,作为下面预测分析的特征[13]。
-
为了确定全脑功能连接是否与这些晚期青少年的健康危险性行为相关,本文使用基于SVM的SVR检验了全脑连接特征是否能用于预测个体的健康危险性行为分数[10]。由于对象有限,使用了留一法交叉验证,即在迭代过程中,留出一个对象的数据作为验证,剩下的n-1个对象的数据作为训练集。每一次迭代包括3步:1)特征选择,在训练集中计算每一条连接与健康危险性行为的皮尔逊相关系数,并使用不同的p阈值选择出与健康危险性有显著相关的边,本文选择了典型的p阈值,即0.005,0.008,0.01,0.015,0.02;2)模型建立,基于选出的连接数据使用支持向量回归的方法训练数据进行多项式核函数拟合;3)预测,把留出的对象数据放入每一个模型以产生一个预测的健康危险性行为分数。所有的迭代完成之后,通过计算所有对象的预测值和实际测量值的皮尔逊相关系数来评估每一种模型的预测能力[14]。
为了进一步研究与HBICA的相关性呈现正的、负的特征网络在对健康危险性行为的预测中所发挥的作用大小,本文根据特征选择符号中的皮尔逊相关系数的符号(正或负)建立了正的特征模型和负的特征模型[14],并分别重复上述迭代过程,通过训练数据分别拟合了两个方向的回归,并对这两个模型下的预测值和实际测量值的皮尔逊相关系数来评估正负特征网络的预测能力。
-
由于留一法交叉验证的属性,在每一次迭代中所选择的功能连接会略有差异,其中出现在每一轮交叉验证里的功能连接被称之为一致性的功能连接[10]。这些一致性功能连接呈现出了对健康危险性行为较强的预测能力。
基于一致功能连接,本文进一步进行网络分析,文献[10]按照功能把全脑分成6个网络,即小脑、带状盖网络、默认网络、额顶网络、枕叶网络和感觉运动网络。为了考察各个网络对预测能力的贡献情况,计算这些网络内边和网络间边的权重,即把与网络相关的连接权重加起来,如果功能连接属于不同网络,则每个相关网络各取权重的一半。
-
作为第一步,本文评估了全脑连接是否能预测晚期青少年的健康危险性行为。基于特征选择阈值P < 0.01,预测和观察值之间的相关系数r=0.388 2 (P = 0.005 8),产生了显著的预测,其他阈值P < 0.008, 0.015下也有显著性,本文选取P < 0.01进行分析[14]。
对正的特征模型,基于特征选择阈值P < 0.01,预测和观察值之间的相关系数r=0.426 2 (P = 0.002 3),对负的特征模型在多项式核函数下没有产生显著的预测,但是负的特征模型在线性核函数下显示出了显著性r=0.446 2 (P =0.001 3)。
-
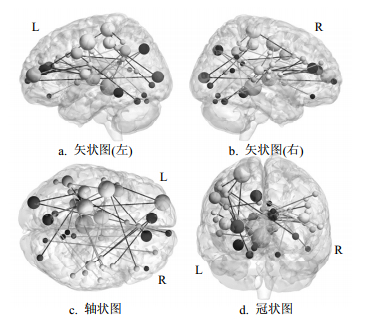
由于留一法交叉验证中,每一次迭代中所选择的训练样本集略有差异,当P < 0.01时,出现在每一轮交叉验证里的一致性功能连接一共有31条(如表 1、图 1所示),可见这31条动态功能连接具有较强的预测能力,其中14条功能连接与健康危险性行为呈正相关,17条功能连接与健康危险性行为呈负相关。
表 1 与HBICA相关的连接
功能连接 MNI坐标 r p 与HBICA正相关的连接 'dFC/med_cerebellum' [-42,7,36]/[-16,-64,-21] 0.578 1 0.000 0 'post_parietal/post_occipital' [-41,-31,48]/[33,-81,-2] 0.530 4 0.000 1 post_occipital/vmPFC' [-29,-88,8]/[6,64,3] 0.529 5 0.000 1 'post_parietal/post_occipital' [-41,-31,48]/[-29,-88,8] 0.515 4 0.000 2 'mid_insula/ACC' [-42,-3,11]/[9,39,20] 0.485 5 0.000 5 'mid_insula/dlPFC' [-42,-3,11]/[46,28,31] 0.480 5 0.000 6 'vFC/vmPFC' [43,1,12]/[-11,45,17] 0.470 0 0.000 9 'parietal/IPL' [-24,-30,64]/[-48,-47,49] 0.453 6 0.001 4 'post_insula/vmPFC' [42,-24,17]/[-11,45,17] 0.452 8 0.001 4 'precentral_gyrus/aPFC' [-54,-9,23]/[-29,57,10] 0.449 6 0.001 6 'parietal/aPFC' [-24,-30,64]/[-29,57,10] 0.448 8 0.001 5 'parietal/IPL' [-24,-30,64]/[-41,-40,42] 0.445 3 0.001 7 'angular_gyrus/basal_ganglia' [-41,-47,29]/[-6,17,34] 0.429 3 0.002 5 'mid_insula/ACC' [33,-12,16]/[-2,30,27] 0.420 7 0.003 1 与HBICA负相关的连接 'post_cingulate/vFC' [-4,-31,-4]/[-48,6,1] -0.515 1 0.000 2 'precentral_gyrus/post_cingulate' [46,-8,24]/[-4,-31,-4] -0.505 2 0.000 3 'IPL/post_cingulate' [-53,-50,39]/[-4,-31,-4] -0.502 0 0.000 3 'precentral_gyrus/thalamus' [-44,-6,49]/[11,-12,6] -0.499 8 0.000 3 'precentral_gyrus/med_cerebellum' [-44,-6,49]/[-11,-72,-14] -0.471 6 0.000 8 'occipital/temporal' [-9,-72,41]/[51,-30,5] -0.469 5 0.000 8 'temporal/occipital' [59,-13,8]/[-9,-72,41] -0.458 4 0.001 2 'post_cingulate/ACC' [10,-55,17]/[9,39,20] -0.439 4 0.002 0 'IPL/aPFC' [54,-44,43]/[-29,57,10] -0.439 3 0.002 0 'precentral_gyrus/thalamus' [-44,-6,49]/[-12,-12,6] -0.434 7 0.002 2 'dFC/TPJ' [-42,7,36]/[-52,-63,15] -0.429 8 0.002 5 'precuneus/occipital' [5,-50,33]/[-28,-42,-11] -0.428 0 0.002 5 'post_insula/thalamus' [42,-24,17]/[-12,-12,6] -0.426 2 0.002 7 'IPL/thalamus' [-53,-50,39]/[-12,-12,6] -0.423 6 0.002 9 'aPFC/med_cerebellum' [27,49,26]/[14,-75,-21] -0.423 5 0.002 9 'precuneus/vlPFC' [9,-43,25]/[46,39,-15] -0.422 6 0.002 9 'precentral_gyrus/occipital' [44,-11,38]/[-28,-42,-11] -0.414 7 0.003 6 为了考察各个网络对健康危险性行为的预测能力情况,本文计算了这些网络内边和网络间边的权重。对每个网络的特征权重计算结果分析发现感觉运动网络,带状盖网络,默认网络和额顶网络相对有较大的总权重,意味着这4个网络对健康危险性行为有较大的影响,如图 2所示。按照正负相关的进一步分析发现在正相关中感觉运动网络相关性最强,而负相关中带状盖网络,默认网络和额顶网络有较强的预测能力。
基于功能连接是属于网络内部的连接还是网络之间的连接进行分类,发现在默认网络内部和带状盖网络内部的连接相对较丰富,且与健康危险性行为呈负相关。其他网络内部连接都比较少,绝大部分连接位于网络之间,且主要呈现为带状盖网络和额顶网络之间的连接,以及感觉运动网络与带状盖网络、默认网络和额顶网络之间的连接呈现出了异常(见图 2和表 1)。
-
不同脑区对预测的贡献大小不同,有些脑区发挥的作用大,有些脑区发挥的作用小。本文利用脑区加权来标示脑区对预测所起的作用,即对所有跟该脑区相连的一致性功能连接进行权重求和并除以2。与一致性功能连接相关的脑区如图 1所示,球的直径大小表示了脑区权重,直径越大,预测能力越强。由图可知几个脑区相比其他脑区直径更大,表明它们具有较强的预测能力,主要是位于带状盖网络的扣带后回(post_cingulate)、丘脑(thalamus)、前额叶腹部(vFC),位于感觉运动网络的中央前回(precentral_gyrus)、顶叶中部及后部(parietal,post_parietal)、脑岛(insula),位于额顶控制网络的前额叶前部(aPFC),前额叶背侧(dFC),顶下小叶(IPL),位于默认网络的前扣带回(ACC),腹内侧前额叶皮层(vmPFC)。
-
本文使用支持向量回归分析发现全脑动态功能连接能较好地用于预测个体的健康危险性行为,可见全脑动态功能连接的波动是一个敏感而有信息量的指标,它提供了静息态下大脑动态组织的重要信息[15]。
同时,也发现了与健康危险性行为相关的功能连接模式。这些连接绝大部分位于网络之间,其中感觉运动网络,带状盖网络,默认网络和额顶网络具有较强的预测能力。带状盖网络和额顶网络是人类两个分开而平行的控制网络,它们之间是一种既分工又合作的关系[16],这两个网络间的连接减弱意味着他们之间平衡性的破坏。这两个网络与感觉运动网络的相关性变弱,可能导致个体对危险性行为的不敏感,或是对行为的控制能力变弱。研究中出现的腹内侧前额叶皮层和扣带回是大脑的默认网络的重要脑区[17],研究认为默认网络执行着一些重要功能,比如情感调整、自我监测[18],默认网络自身连接的减弱和跟外界连接的改变可能反应了在自适应调整情感和自我监测上出现了问题。可能正是由于这些网络内部和网络之间连接出现了问题,从而导致了个体高危险性行为的发生。
Prediction of the Health-Risk Behavior by Using Dynamic Functional Connectivity
-
摘要: 为了研究健康危险性行为的脑网络特征,该文采集了49个被试的静息态功能磁共振数据。使用每一个对象动态功能连接网络的低频振荡振幅作为特征,利用支持向量回归对个体的健康危险行为进行预测。结果表明动态功能连接能较好地预测健康危险性行为特征,并提取了与之相关的功能连接模式,对预测有重要作用的连接绝大部分位于网络之间,且主要呈现为带状盖网络和额顶网络之间的连接,以及感觉运动网络与它们之间的连接相关。Abstract: In order to investigate the brain network characteristics of the health-risk behavior, we collected fMRI data of 49 subjects under rest state. The fluctuation amplitude of dynamic functional connectivity is used as the features of support vector regression (SVR) to predict the health-risk behavior. The results show a good correlation between spontaneous fluctuation of rest state and the health-risk behavior. Some informational functional connectivities could be used to predict the health-risk behavior and they mainly locate among the connections of networks:mainly cingulo-opercular network, frontoparietal network, sensorimotor network, etc..
-
表 1 与HBICA相关的连接
功能连接 MNI坐标 r p 与HBICA正相关的连接 'dFC/med_cerebellum' [-42,7,36]/[-16,-64,-21] 0.578 1 0.000 0 'post_parietal/post_occipital' [-41,-31,48]/[33,-81,-2] 0.530 4 0.000 1 post_occipital/vmPFC' [-29,-88,8]/[6,64,3] 0.529 5 0.000 1 'post_parietal/post_occipital' [-41,-31,48]/[-29,-88,8] 0.515 4 0.000 2 'mid_insula/ACC' [-42,-3,11]/[9,39,20] 0.485 5 0.000 5 'mid_insula/dlPFC' [-42,-3,11]/[46,28,31] 0.480 5 0.000 6 'vFC/vmPFC' [43,1,12]/[-11,45,17] 0.470 0 0.000 9 'parietal/IPL' [-24,-30,64]/[-48,-47,49] 0.453 6 0.001 4 'post_insula/vmPFC' [42,-24,17]/[-11,45,17] 0.452 8 0.001 4 'precentral_gyrus/aPFC' [-54,-9,23]/[-29,57,10] 0.449 6 0.001 6 'parietal/aPFC' [-24,-30,64]/[-29,57,10] 0.448 8 0.001 5 'parietal/IPL' [-24,-30,64]/[-41,-40,42] 0.445 3 0.001 7 'angular_gyrus/basal_ganglia' [-41,-47,29]/[-6,17,34] 0.429 3 0.002 5 'mid_insula/ACC' [33,-12,16]/[-2,30,27] 0.420 7 0.003 1 与HBICA负相关的连接 'post_cingulate/vFC' [-4,-31,-4]/[-48,6,1] -0.515 1 0.000 2 'precentral_gyrus/post_cingulate' [46,-8,24]/[-4,-31,-4] -0.505 2 0.000 3 'IPL/post_cingulate' [-53,-50,39]/[-4,-31,-4] -0.502 0 0.000 3 'precentral_gyrus/thalamus' [-44,-6,49]/[11,-12,6] -0.499 8 0.000 3 'precentral_gyrus/med_cerebellum' [-44,-6,49]/[-11,-72,-14] -0.471 6 0.000 8 'occipital/temporal' [-9,-72,41]/[51,-30,5] -0.469 5 0.000 8 'temporal/occipital' [59,-13,8]/[-9,-72,41] -0.458 4 0.001 2 'post_cingulate/ACC' [10,-55,17]/[9,39,20] -0.439 4 0.002 0 'IPL/aPFC' [54,-44,43]/[-29,57,10] -0.439 3 0.002 0 'precentral_gyrus/thalamus' [-44,-6,49]/[-12,-12,6] -0.434 7 0.002 2 'dFC/TPJ' [-42,7,36]/[-52,-63,15] -0.429 8 0.002 5 'precuneus/occipital' [5,-50,33]/[-28,-42,-11] -0.428 0 0.002 5 'post_insula/thalamus' [42,-24,17]/[-12,-12,6] -0.426 2 0.002 7 'IPL/thalamus' [-53,-50,39]/[-12,-12,6] -0.423 6 0.002 9 'aPFC/med_cerebellum' [27,49,26]/[14,-75,-21] -0.423 5 0.002 9 'precuneus/vlPFC' [9,-43,25]/[46,39,-15] -0.422 6 0.002 9 'precentral_gyrus/occipital' [44,-11,38]/[-28,-42,-11] -0.414 7 0.003 6 -
[1] 季成叶.青少年健康危险行为[J].中国学校卫生, 2007, 28(4):289-291. doi: 10.3969/j.issn.1000-9817.2007.04.001 JI Chen-ye. The health-risk behavior of adolescents[J].Chinese Journal of School Health, 2007, 28(4):289-291. doi: 10.3969/j.issn.1000-9817.2007.04.001 [2] WANG M, YI J, CAI L, et al. Development and psychometric properties of the health-risk behavior inventory for Chinese adolescents[J]. BMC Med Res Methodol, 2012, 12:94. doi: 10.1186/1471-2288-12-94 [3] WELTE J W, WIECZOREK W F, BARNES G M, et al. The relationship of ecological and geographic factors to gambling behavior and pathology[J]. J Gambl Stud, 2004, 20(4):405-423. doi: 10.1007/s10899-004-4582-y [4] JESSOR R. Risk behavior in adolescence:a psychosocial framework for understanding and action[J]. Developmental Review, 1992, 12(4):374-390. doi: 10.1016/0273-2297(92)90014-S [5] JIANG W, SHI F, LIAO J, et al. Disrupted functional connectome in antisocial personality disorder[J]. Brain Imaging Behav, 2016, doi: 10.1007/s11682-016-9572-z. [6] HUTCHISON R M, WOMELSDORF T, GATI J S, et al. Resting-state networks show dynamic functional connectivity in awake humans and anesthetized macaques[J]. Hum Brain Mapp, 2013, 34(9):2154-2177. doi: 10.1002/hbm.v34.9 [7] ALLEN E A, DAMARAJU E, PLIS S M, et al. Tracking whole-brain connectivity dynamics in the resting state[J]. Cereb Cortex, 2014, 24(3):663-676. doi: 10.1093/cercor/bhs352 [8] HUTCHISON R M, MORTON J B. Tracking the brain's functional coupling dynamics over development[J]. J Neurosci, 2015, 35(17):6849-6859. doi: 10.1523/JNEUROSCI.4638-14.2015 [9] SHEN H, WANG L, LIU Y, et al. Discriminative analysis of resting-state functional connectivity patterns of schizophrenia using low dimensional embedding of fMRI[J]. NeuroImage, 2010, 49:3110-3121. doi: 10.1016/j.neuroimage.2009.11.011 [10] DOSENBACH N U, NARDOS B, COHEN A L, et al. Prediction of individual brain maturity using fMRI[J]. Science, 2010, 329(5997):1358-1361. doi: 10.1126/science.1194144 [11] YU R, CHIEN Y L, WANG H L, et al. Frequency-specific alternations in the amplitude of low-frequency fluctuations in schizophrenia[J]. Hum Brain Mapp, 2014, 35(2):627-637. doi: 10.1002/hbm.v35.2 [12] LEONARDI N, VAN DE VILLE D. On spurious and real fluctuations of dynamic functional connectivity during rest[J]. Neuroimage, 2015, 104:430-436. doi: 10.1016/j.neuroimage.2014.09.007 [13] SHEN H, LI Z, QIN J, et al. Changes in functional connectivity dynamics associated with vigilance network in taxi drivers[J]. Neuroimage, 2016, 124:367-378. doi: 10.1016/j.neuroimage.2015.09.010 [14] FINN E S, SHEN X, SCHEINOST D, et al. Functional connectome fingerprinting:identifying individuals using patterns of brain connectivity[J]. Nat Neurosci, 2015, 18(11):1664-1671. doi: 10.1038/nn.4135 [15] QIN J, CHEN S G, HU D, et al. Predicting individual brain maturity using dynamic functional connectivity[J]. Front Hum Neurosci, 2015, 9:418. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=Doaj000004205449 [16] FAIR D A, DOSENBACH N U, CHURCH J A, et al. Development of distinct control networks through segregation and integration[J]. Proc Natl Acad Sci USA, 2007, 104(33):13507-13512. doi: 10.1073/pnas.0705843104 [17] SYLVESTER C M, CORBETTA M, RAICHLE M E, et al. Functional network dysfunction in anxiety and anxiety disorders[J]. Trends Neurosci, 2012, 35(9):527-535. doi: 10.1016/j.tins.2012.04.012 [18] BUCKNER R L, ANDREWS-HANNA J R, SCHACTER D L. The brain's default network:anatomy, function, and relevance to disease[J]. Ann N Y Acad Sci, 2008, 1124:1-38. doi: 10.1196/annals.1440.011 -

 ISSN
ISSN 




 下载:
下载: